All of our recommendations, unless specified, relate to acute COVID-19 in adults.
Some of our recommendations vary according to the severity of COVID-19 illness. Definitions of the categories are based on World Health Organization (WHO) criteria and can be viewed by clicking the plus (+) signs below.
RECOMMENDATION: We recommend use of Remdesivir in hospitalized patients with moderate to critical COVID-19 (conditional recommendation), probably reduce mortality, time to clinical improvement and progression to NIV or High Flow Nasal oxygen (HFNO).
DATE OF RECOMMENDATION: 16th June 2022
A conditional recommendation is one for which the panel is less confident about the balance between desirable and undesirable effects of the intervention, or other aspects, such as cost and feasibility.
Definition of moderate illness:
- Pneumonia (clinical or radiological) OR hypoxia (SpO2 <94% in adults with no underlying lung disease)
- AND respiratory rate ≤30/min
- AND SpO2 ≥90% on room air
Definition of severe:
Pneumonia with ANY ONE of the following:
- Respiratory rate >30/min
- Severe respiratory distress
- SpO2 <90% on room air
- NO invasive or non-invasive respiratory support needed
Definition of critical:
- Requirement for high-level respiratory support: non-invasive ventilation, high-flow oxygen (≥20 litres per minute) or invasive mechanical ventilation
- OR acute respiratory distress syndrome (PaO2/FiO2 ratio of <300)
- OR sepsis
- OR shock
It has long been suggested that Remdesivir an antiviral is likely to be most beneficial in the early stages of the disease before the hyperinflammatory phase has set in, and hence should preferably be given early in the course of the disease with symptom onset less than 5 days. However, till recently this effect was not demonstrable.
Updation of recommendations with the findings from few new trials and final results of the Solidarity trial revealed that Remdesivir probably reduces all-cause mortality, time to clinical improvement, progression to NIV/HFNO, secondary infections and any adverse events. Remdesivir may reduce thrombotic complications, time to clinical recovery, progression to oxygen therapy, non-invasive, invasive and ECMO in hospitalized COVID-19 patients. The evidence was very uncertain for progression to invasive mechanical ventilation (IMV), serious adverse events and adverse event leading to drug interruption.
The earlier trials were conducted when steroids were not the standard of care, and other respiratory treatment protocols were unclear, which may have impacted the benefit of Remdesivir. However, with steroids becoming the standard of care and improved respiratory therapy protocols with regard to non invasive ventilation and invasive mechanical ventilation in hypoxic hospitalized patients, the overall data now indicates that Remdesivir provides a benefit over and above that demonstrated by steroids in those who have moderate to critical COVID-19. Though the mortality benefit of 10% is not dramatic, the benefit of 28% in prevention of progression to high flow nasal oxygen or NIV is indeed remarkable and will likely help with de congestion of hospitals and more beds and treatment modalities being available to deserving patients.
Date of latest search: 04th May 2022
Date of completion & presentation to Anti-Viral Expert Working Group: 07th June 2022
Date of planned review: January 2023
Evidence synthesis team: Naveena G Princy ,Hanna Alexander, Anupa Thampy, Richard Kirubakaran, Bhagteshwar Singh and Priscilla Rupali.
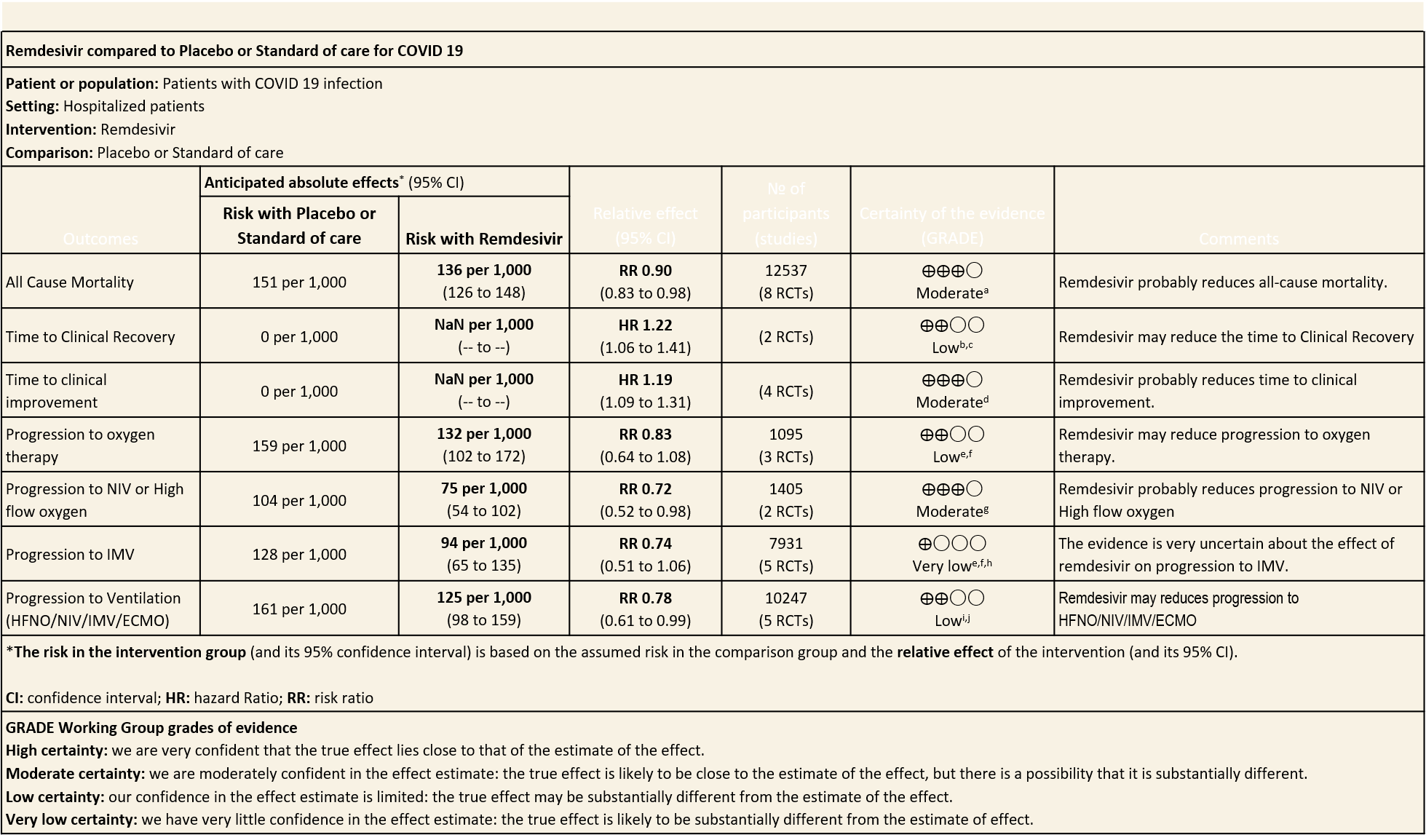
Summary of Findings Table 1
Explanations
a. Downgraded by one level for serious risk of bias; Spinner 2020, Mahajan, and Karim Ali 2022 has a high risk of bias and two studies reported Some concerns.
b. Downgraded by one level of serious risk of bias; Spinner 2020 was at high risk of bias and contributed to 36.1% of the weight in the meta-analysis
c. Downgraded by one level for serious indirectness; Spinner 2020 has moderate severity of patients only and ACTT Beigel 2020 had a mixture of patients of different severity.
d. Downgraded by one level of serious risk of bias; Spinner 2020 was at high risk of bias and contributed 17.3% and Ader F 2021 contributed to 33.5% of the weight in the meta-analysis and reported some concerns in risk of bias
e. Downgraded by one level for serious risk of bias; Karim Ali 2021 has a high risk of bias and AderF 2021 Elsalam 2021 has some concerns;
f. Downgraded by one level for serious imprecision: the 95% CI includes both benefit and harm
g. Downgraded to one level for serious imprecision; RR 0.72 95% CI is 0.52 - 0.98 and it does not meet the Optimal information size (OIS) criteria
h. Downgraded by one level for serious inconsistency: The I 2 is 82%
i. Downgraded by one level for serious risk of bias; Mahajan 2021 has a high risk of bias and AderF 2021 Elsalam 2021 has some concerns
j. Downgraded by one level for serious inconsistency: The I 2 is 63 %
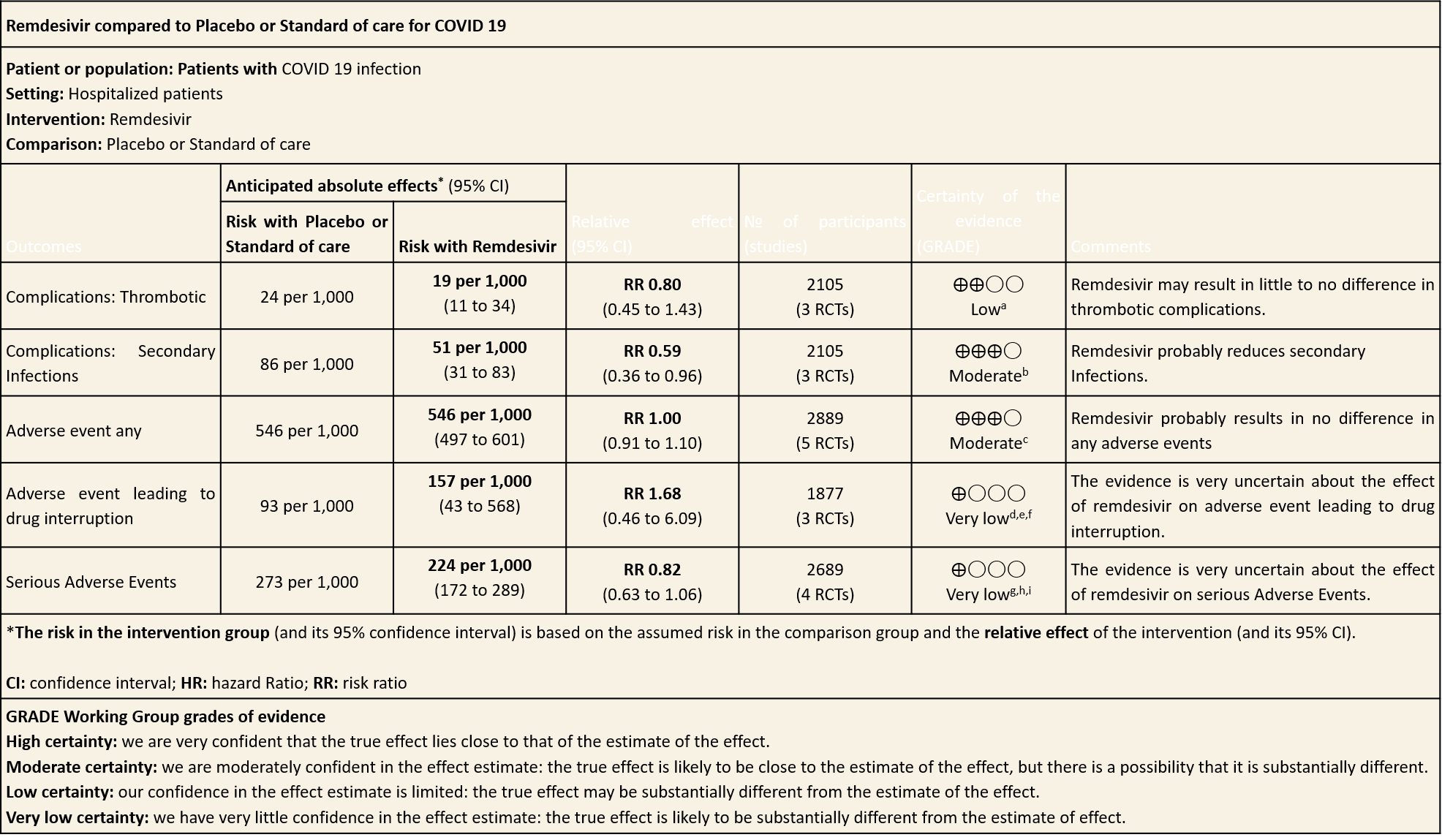
Summary of Findings Table 2
Explanations
a. Downgraded by two levels for serious imprecision; the number of events were few and the 95% CI of the effect estimate included clinically important reductions and increases in thrombotic events with Remdesivir. OIS criteria not met
b. Downgraded to one level for serious imprecision; RR 0. 59 (95% CI is 0.36 - 0.96) and it does not meet the Optimal information size (OIS) criteria
c. Downgraded by one level of serious risk of bias; Spinner 2020 was at high risk of bias and contributed to 19.4% and Ader 2021 has some concerns which contributed to 30.3% of the weight in the meta-analysis and Elsalam 2021 also reported some concerns
d. Downgraded by one level of serious risk of bias; Spinner 2020 was at high risk of bias
e. Downgraded by one level for serious inconsistency: The I 2 is 76%
f. Downgraded by two levels for serious imprecision: the 95% CI is wide (0.46 to 6.09)
g. Downgraded by one level of serious risk of bias; Spinner 2020 was at high risk of bias and contributed to 12.4% and Ader 2021 has some concerns which contributed to 35.5% of the weight in the meta-analysis
h. Downgraded by one level for serious inconsistency: The I 2 is 63%
i. Downgraded by one level for serious imprecision: the 95% CI is wide (0.63-1.06)
Remdesivir is a prodrug that is converted to an active triphosphate form that inhibits viral RNA production through RNA dependent RNA polymerase.(1). It was developed as a therapeutic agent for treating RNA-based viruses such as Ebola virus, MERS, SARS-CoV-1 and other zoonotic coronaviruses (2). It has demonstrated in vitro and in vivo antiviral activity against SARS-CoV-2 (3).
Remdesivir is widely used across the world and a number of guidelines have recommended its use in hospitalized hypoxic COVID-19 patients (4). The WHO recently recommended the use of Remdesivir with caution in individuals with non-severe COVID-19 who are at high risk of hospitalisation(5). It is also approved by the FDA for the treatment of COVID-19 in nonhospitalized and hospitalised paediatric patients weighing 3.5 kg to 40 kg or <12 years and weighing ≥3.5 kg(6).
Currently Remdesivir is a widely available antiviral drug in the country. The Ministry of Health & Family Welfare recommends Remdesivir for moderate to severe COVID-19 hospitalized patients on supplemental oxygen (7) as it appears to have a relatively good safety profile.
This review aims to provide a summary of the available evidence from randomized clinical trials of Remdesivir for treatment of acute COVID-19, for any duration, which could guide clinicians and researchers regarding the appropriate use of this drug in the future.
We searched the Cochrane Central Register of Controlled Trials (CENTRAL), Epistemonikos, and the COVID‐19‐specific resource www.covid‐nma.com, for studies of any publication status and in any language. We also reviewed reference lists of systematic reviews and included studies. We performed all searches up to 4th May 2022. We included randomized controlled trials (RCTs) testing Remdesivir in people with COVID‐19, and extracted data for the following pre-defined outcomes:
Critical (primary for this review):
-
- Mortality (all-cause): at 28-30 days, or in-hospital
- Progression to:
- Oxygen therapy
- Ventilation: non-invasive or invasive
- Critical/Intensive care (any reason) - Time to clinical improvement
- Time to clinical recovery
- Adverse events: all and serious
Important (secondary):
-
- Time to negative PCR for SARS-CoV-2
- Duration of hospitalization
- Complications of COVID-19:
- Thrombotic events
- Pulmonary function/fibrosis
- Long COVID/post-acute sequelae COVID
- Secondary infections
Two reviewers independently assessed eligibility of search results. One reviewer extracted data from each included study, and assessed risk of bias using the Cochrane Risk of bias (RoB) v2.0 tool. Data and RoB assessments were checked by the second reviewer. The entire RoB assessment was scrutinized by the whole team for this review, to reach consensus.
We used RevMan 5.4 to perform meta‐analysis using a random‐effects model for outcomes where pooling of effect estimates was appropriate. We used risk ratios (RR) for dichotomous outcomes and hazard ratios for continuous outcomes, with 95% confidence intervals (CIs). We performed a subgroup analysis to explore the effect on mortality stratified by different oxygen and ventilation requirements at baseline.
We used the I2 statistic to measure residual heterogeneity. We used GRADE methodology to assess the certainty in the evidence, and documented this in a ‘Summary of findings’ table using GradeProGDT.
We included eight trials involving 12,537 participants, all of whom were adults, and 6408 of whom received Remdesivir(8–15). Four trials were reported, one each from China(9),Canada (12),Egypt (13)and India (11); three from multiple countries in North America, Europe and Asia (8,10,14); one recruited worldwide (15). All trials were done in hospitalized patients. Disease severity, prevalence of comorbidities, and use of co‐interventions (e.g., steroids) varied substantially between trials. Out of the eight trials, three (8,9,15) were found to have low risk of bias across all domains for all outcomes and three(10–12) had high risk of bias across multiple domains. Risk of bias for each domain per trial is displayed alongside the forest plots below. WHO solidarity trial’s interim report has been added for additional data(16).
The following comparisons were investigated in the trials (we compared outcomes for arms randomized to Remdesivir vs. outcomes for arms randomized to standard of care or placebo):
Two trials(8,9) compared Remdesivir with placebo (1299 participants) whereas, six trials (10–15) compared Remdesivir with standard of care (8414 participants). Of these, one trial (10) had two arms with different duration of Remdesivir (5 days arm and 10 days arm) compared with a standard of care arm (596 participants).
Our expert working group classified progression to oxygen, non-invasive ventilation (NIV) and invasive mechanical ventilation (IMV) as critical outcomes and mortality, time to clinical improvement and thrombotic or secondary infections as important outcomes. However, as the situation in the country evolved, the guidelines group upgraded mortality, time to clinical recovery and time to clinical improvement as critical outcomes, as a benefit observed in these outcomes, could reduce pressure on the overwhelmed health system.
Critical (primary) Outcomes
As presented in the ‘Summary of findings’ table, the evidence is moderate for all-cause mortality, time to clinical improvement, progression to NIV/HFNO, secondary infections and any adverse event. The evidence is low for time to clinical recovery, progression to oxygen therapy, progression to ventilation (NIV/HFNO/ECMO/IMV) and thrombotic complications. Also we found very low certainty for progression to IMV, adverse events leading to drug discontinuation and serious adverse events.
(a) All-cause mortality: Moderate certainty of evidence in 12537 patients in eight RCTs (8–15) found that Remdesivir probably reduces mortality when compared to placebo or standard of care (RR 0.90; 95% CI 0.83 to 0.98)).
(b) Time to clinical recovery (time to achieve WHO score 1,2,3 or not requiring hospitalization for oxygen or medical care): Low certainty evidence In 1459 patients from two RCTs (8,10), revealed that Remdesivir may reduce time to clinical recovery in participants receiving Remdesivir compared with those receiving placebo or standard of care; Hazard ratio (HR) was 1.22 (95% CI 1.06 to 1.41). The median time reported in two studies ranged from 6-10 days in the Remdesivir group vs 7-15 days in the control groups (See Results Table 1 for results from each trial). A mean difference to quantify the magnitude of potential benefit was not available.
(c) Time to clinical improvement (>2 point reduction in the WHO ordinal score):
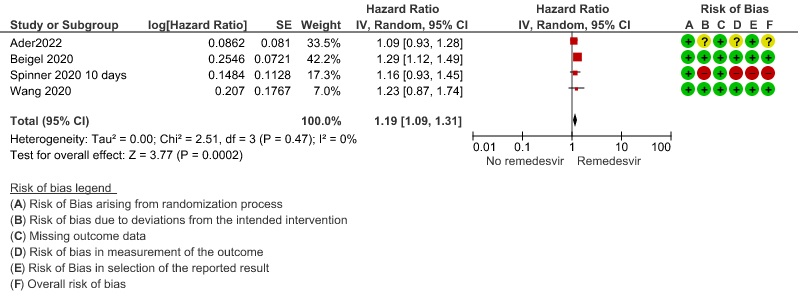
Moderate certainty evidence from in four RCTs (8–10,14) suggested that Remdesivir probably decreases time to clinical improvement (HR 1.19; 95% CI 1.09 to 1.31). The median and IQR ranged from 6-21 days in the Remdesivir group vs. 6-23 days in the control group. (See Results Table 1 for results from each trial). A mean difference to quantify the magnitude of potential benefit was not available.
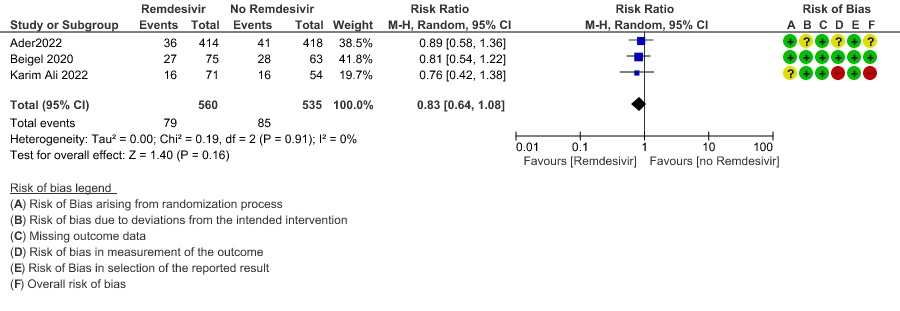
(d) Progression to oxygen therapy: Low certainty evidence in 1095 patients from three RCTs(8,12,14) found that Remdesivir may reduce progression to oxygen therapy vs. standard of care (SOC)/placebo (RR 0.83 ; 95% CI 0.64 to 1.08 ).
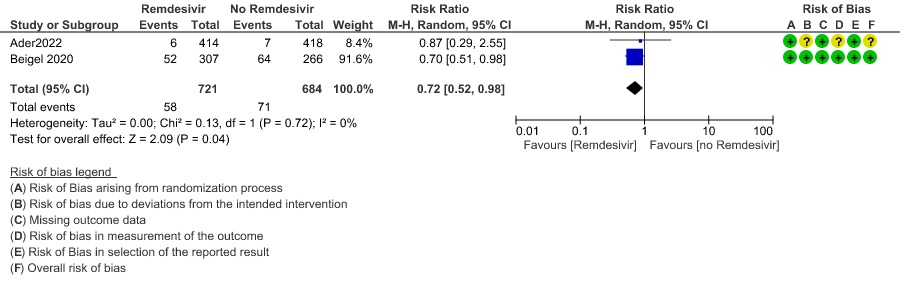
(e) Progression to non-invasive ventilation (NIV) or high flow oxygen (HFO): Moderate certainty evidence from 1405 patients in two RCTs(8,14) found that Remdesivir probably reduces progression to NIV or HFO (RR 0.72; 95% CI; 0.52 to 0.98 ) and varied as widely as 2% to 48% with small sample size and few events.
(f) Progression to invasive mechanical ventilation:Very low certainty evidence from 7931 patients in five RCTs (8,12–15) found an uncertain effect of Remdesivir on progression to invasive mechanical ventilation (RR 0.74 ; 95% CI 0.51 to 1.06 ).
(g) Progression to ventilation (NIV/HFNO/IMV/ECMO): Low certainty evidence from 10247 patients in five RCTs(8,9,11,14,16) found that Remdesivir may reduce progression to ventilation (NIV/HFNO/IMV/ECMO) (RR 0.78; 95% CI 0.61 to 0.99).
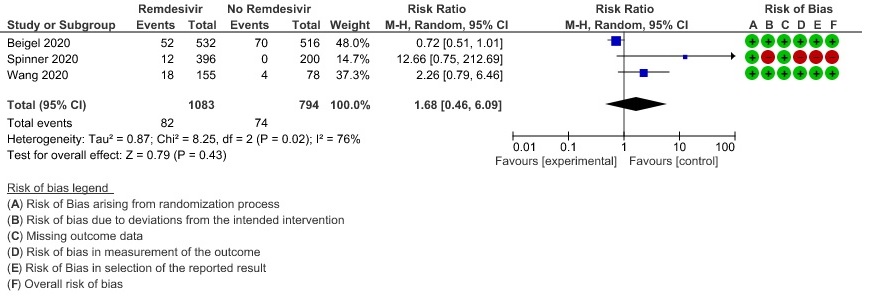
(h) Adverse events: Moderate certainty evidence from 2889 patients in five RCTs (3,8,10,13,14) revealed Remdesivir probably contribute to few or no additional adverse events as compared to placebo (RR 1.00; 95% CI 0.90 to 1.10 ). In addition, very low certainty evidence from 1877 patients in three RCTs (3,8,10) found an uncertain effect of Remdesivir on drug discontinuation due to adverse effects (RR1.68; 95% CI 0.46 to 6.09) and serious adverse events from 2689 patients in four RCTs (8–10,14) [RR 0.82;95% CI 0.63-1.06].
(i) Duration of hospitalization: This was reported in four studies as a median and hence a meta-analysis could not be performed. Wang 2020 (9)] reported a median (IQR) of 21 (12 to 31) days in the Remdesivir group vs. 21 (13.5 to 28.5) days in the SOC or placebo groups. ACTT Beigel 2020 (8)reported median (IQR) as 12 (6-28) days vs. 17 (8-28) days in the SOC/placebo groups.Abd-Elsalam 2022 (13)reported median (IQR) as 10(8-13.5) days vs. 16(12-21) days in the SOC/placebo groups. Ali K 2022 (12) reported median (IQR) as 10(6-18) days vs. 9(6-17) days in the SOC/placebo groups.
Important (secondary) outcomes:
(1) Thrombotic events: Low certainty evidence from three RCTs (8,9,14) in 2105 participants suggests that Remdesivir may make little to no difference to occurrence of thrombotic events when compared to placebo or SOC (RR 0.80; 95% CI 0.45 to 1.43)
(2) Secondary infections: Moderate certainty evidence from three RCTs (8,9,14) in 2105 participants suggests that Remdesivir may reduce secondary infections when compared to placebo or SOC (RR ; 95% CI 0.36 to 0.96 ).
No comparative data could be extracted for time to negative PCR; or post-acute COVID-19 pulmonary function/fibrosis or other sequelae.
Subgroup analysis
A subgroup analysis was attempted as pre-specified by the Expert Working Group.
In three RCTs (3,8,12) with 1332 participants, in the subgroup with low flow oxygen it did show a mortality benefit (RR 0.57; 95% CI 0.34 to 0.95).
In one RCT (9) Remdesivir showed no mortality benefit when participants were stratified by shorter duration of symptoms (<10 days; RR 0.76; 95% CI 0.29 to 1.95) or ≥10 days of symptoms (RR 1.48; 95% CI 0.45 to 4.88).
Disaggregated data could not be obtained for co-morbidities, inflammatory markers or different age groups. Subgroups of pregnancy, lactation, renal failure, liver failure were excluded from most trials. No data were available separately for immunocompromised individuals in any of the trials.
Table 1: Time to clinical improvement and recovery
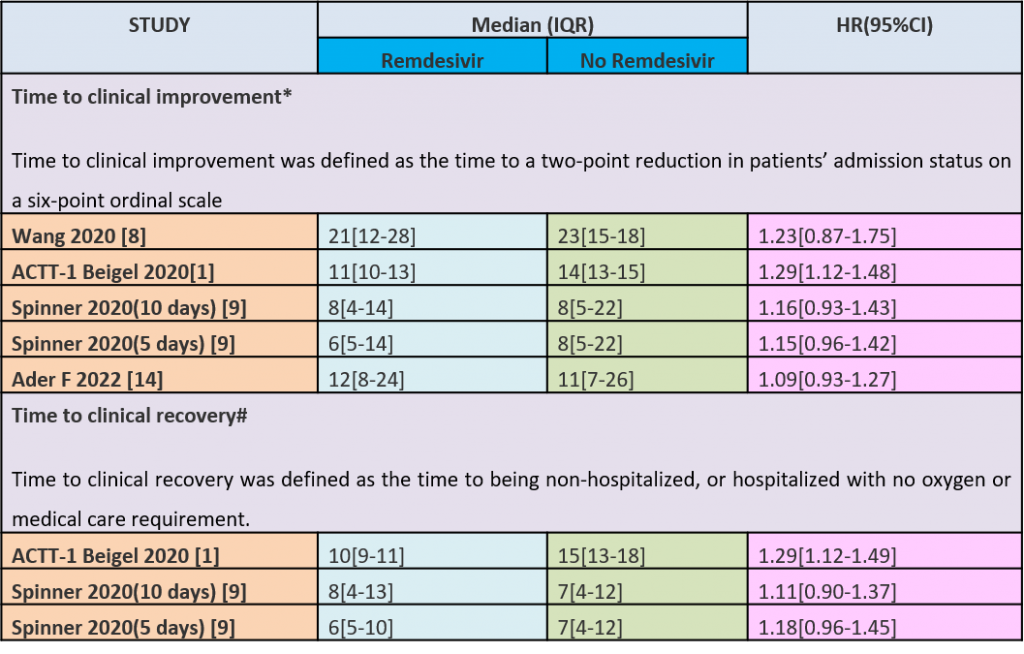
Table 2 Duration of Hospitalization

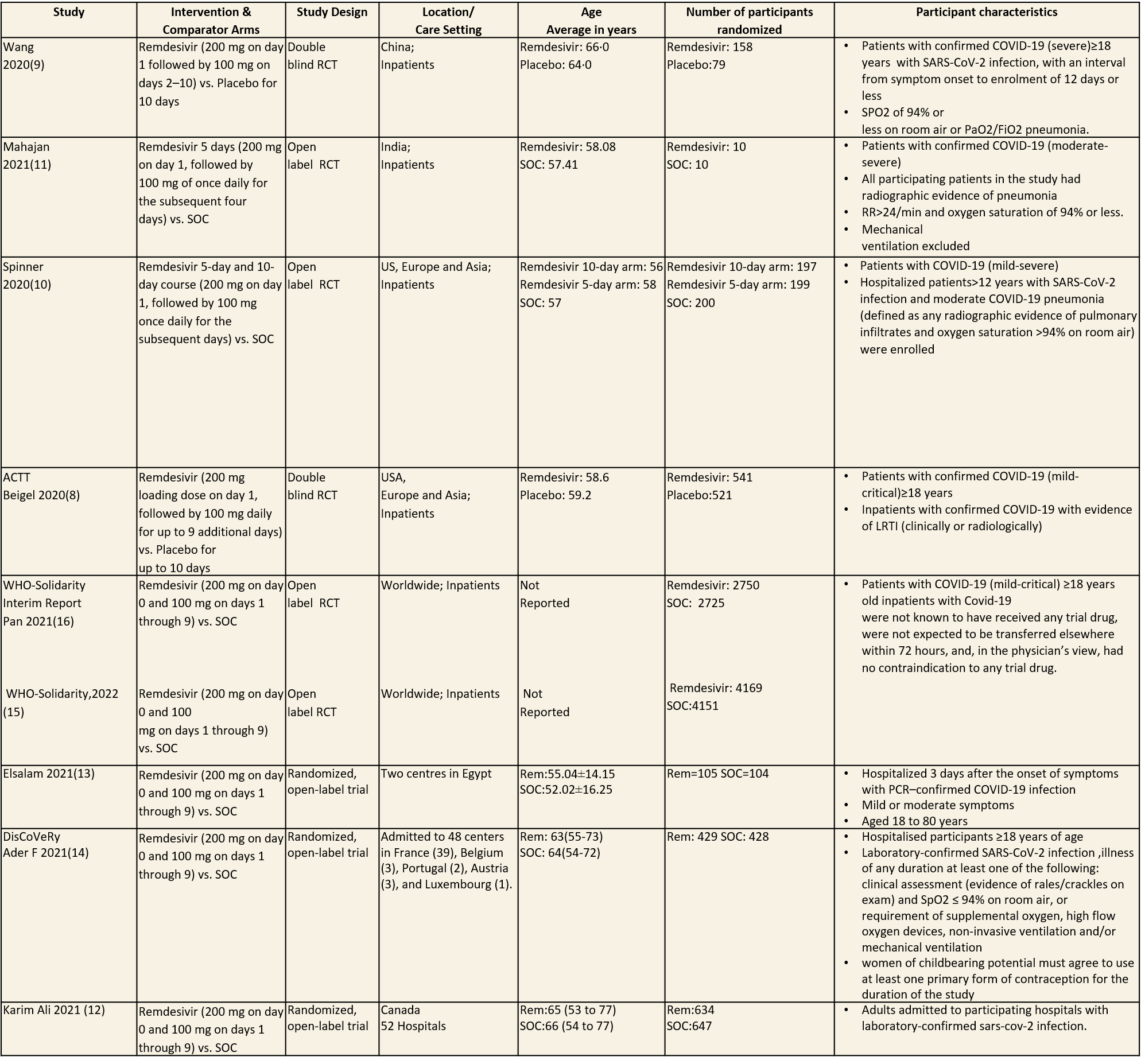
Inclusion and exclusion criteria for the included trials
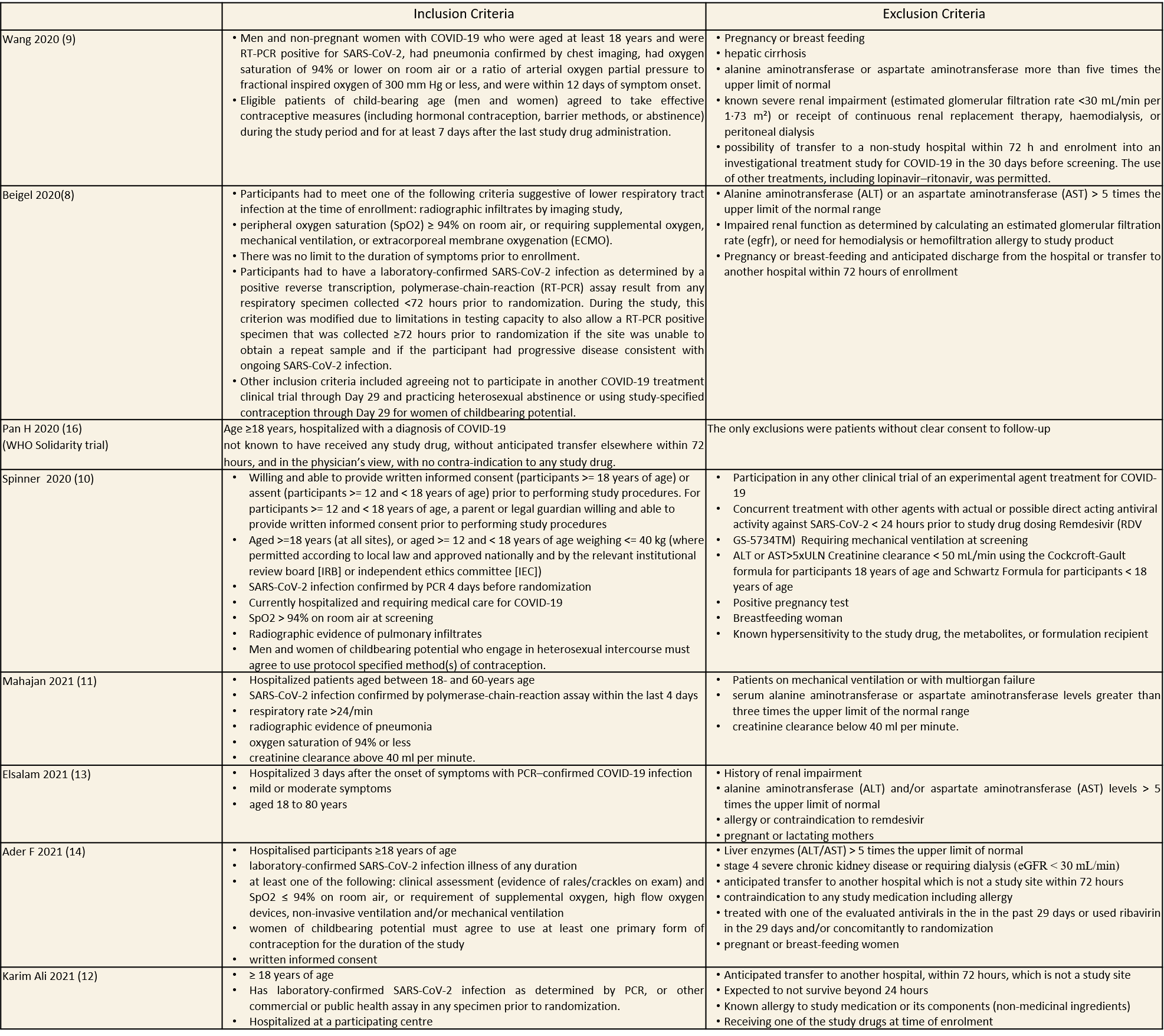
1.All Cause Mortality
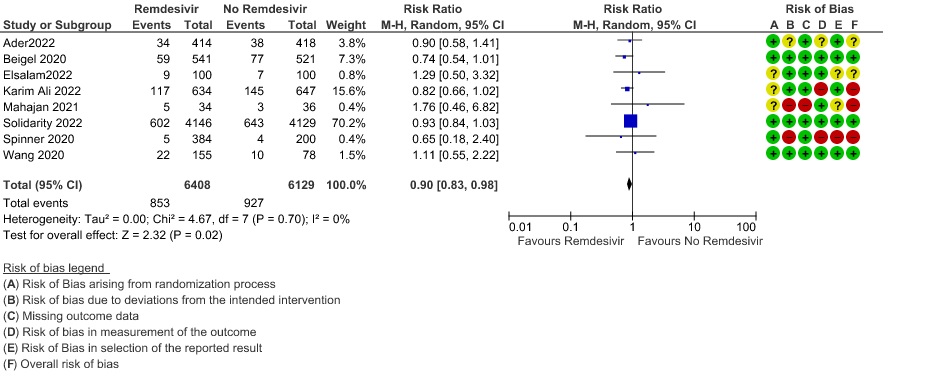
1a. All Cause Mortality (Subgroup )
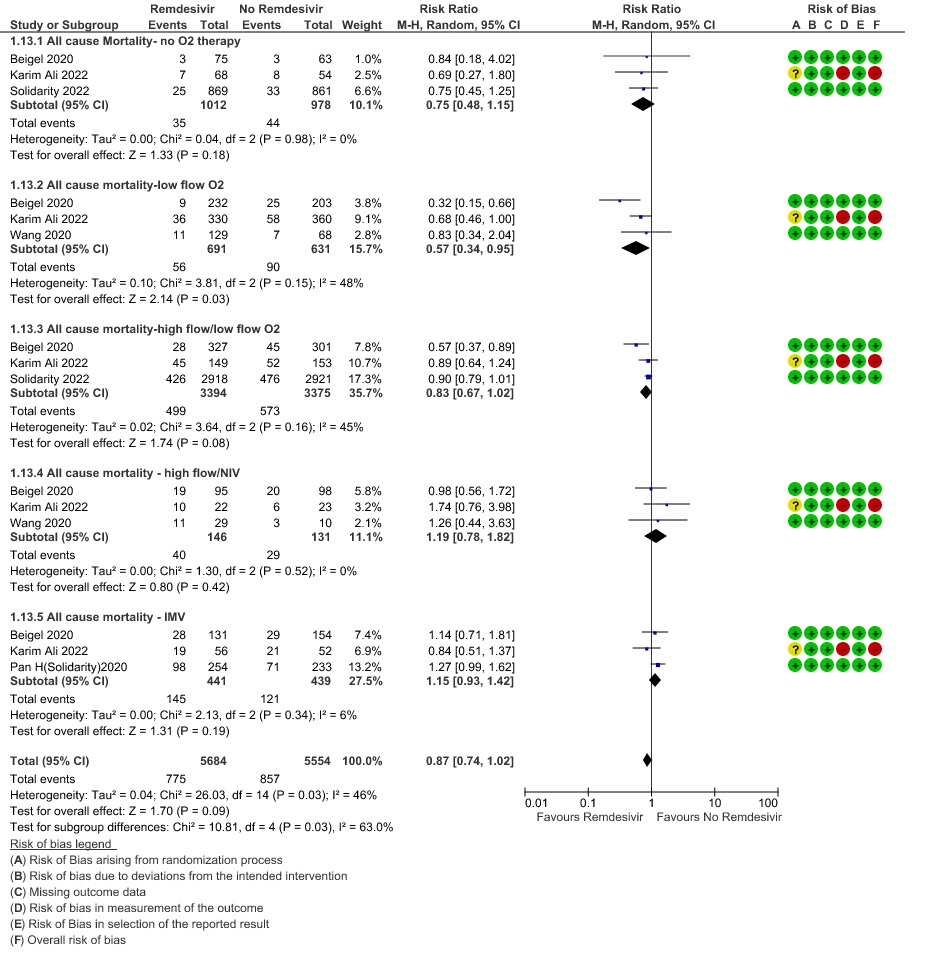
2. Time to clinical recovery
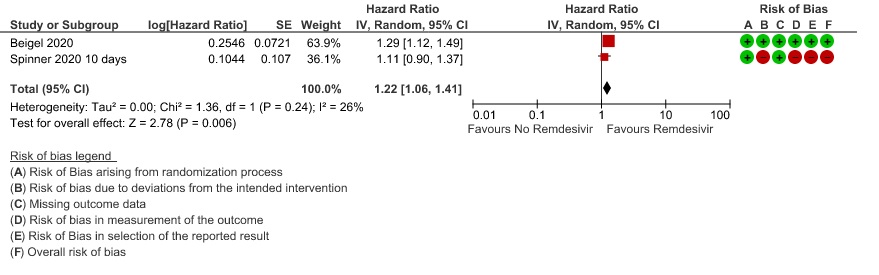
3. Time to clinical Improvement
4. Progression to Oxygen Therapy
5. Progression to non-invasive ventilation/ High low oxygen
6. Progression to Invasive Mechanical Ventilation
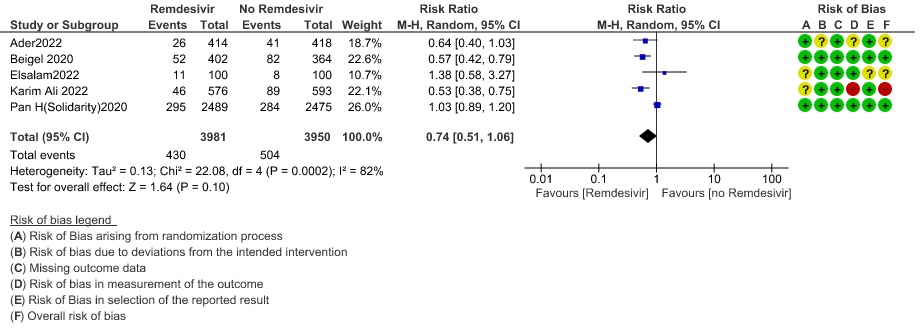
7. Progression to Ventilation (NIV/IMV/HFNO/ECMO)
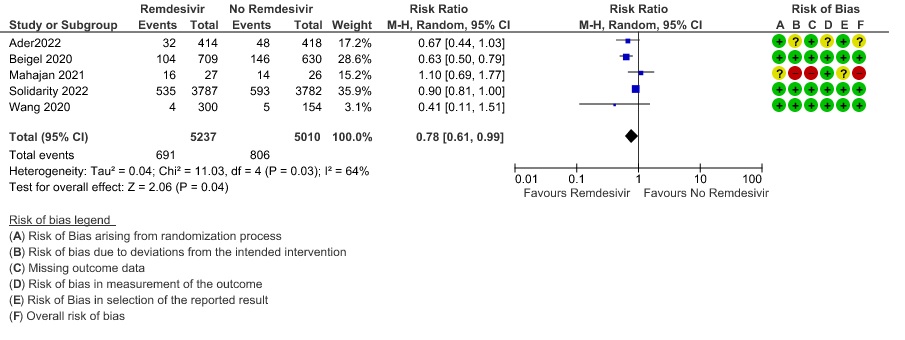
8. Complications – Thrombotic events
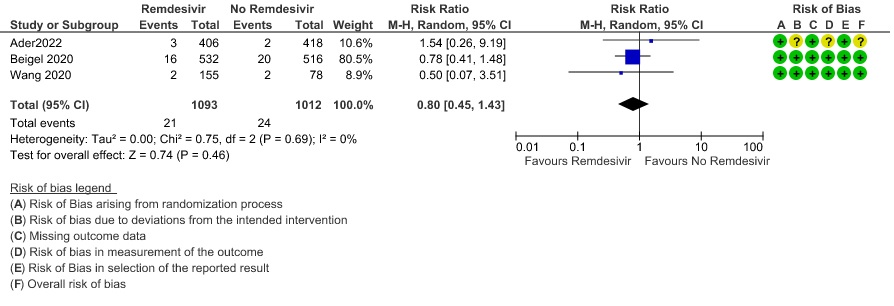
9. Complications – Secondary Infections
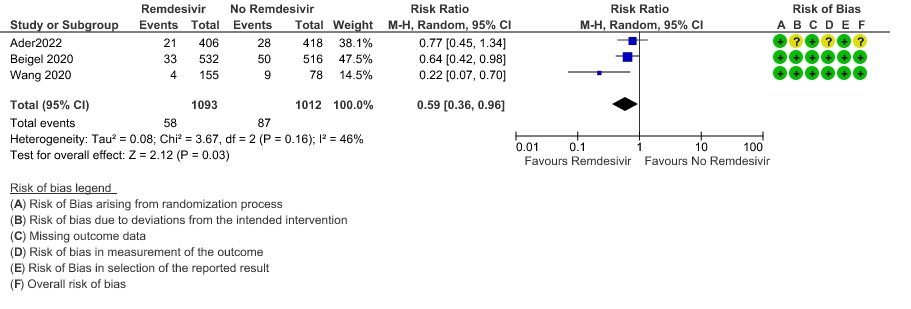
10. Any adverse events
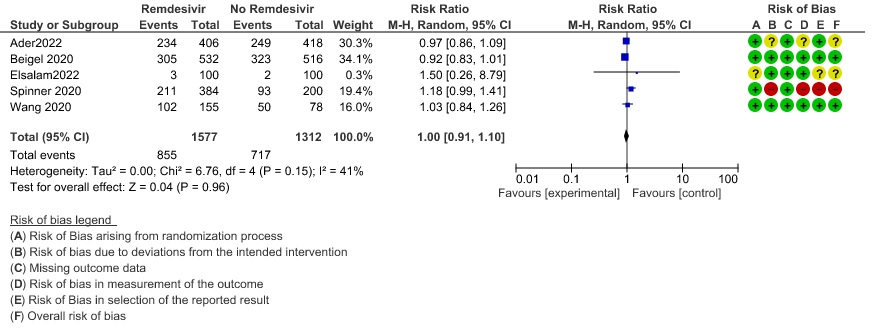
11. Serious Adverse events
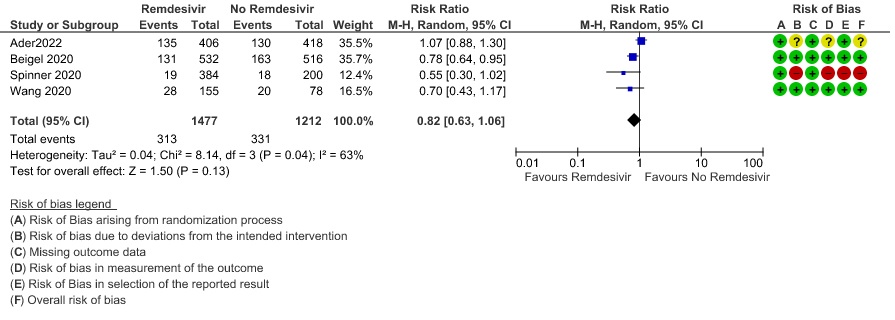
12. Adverse event leading to drug interruption
The Antiviral Expert Working Group met on 07th June 2022 to consider Remdesivir as a treatment for COVID-19. Conflict of interest declarations were reviewed by the Steering Committee; none were found to be relevant to remdesivir.
A summary and then more detailed explanations of the Expert Working Group's judgements follow.
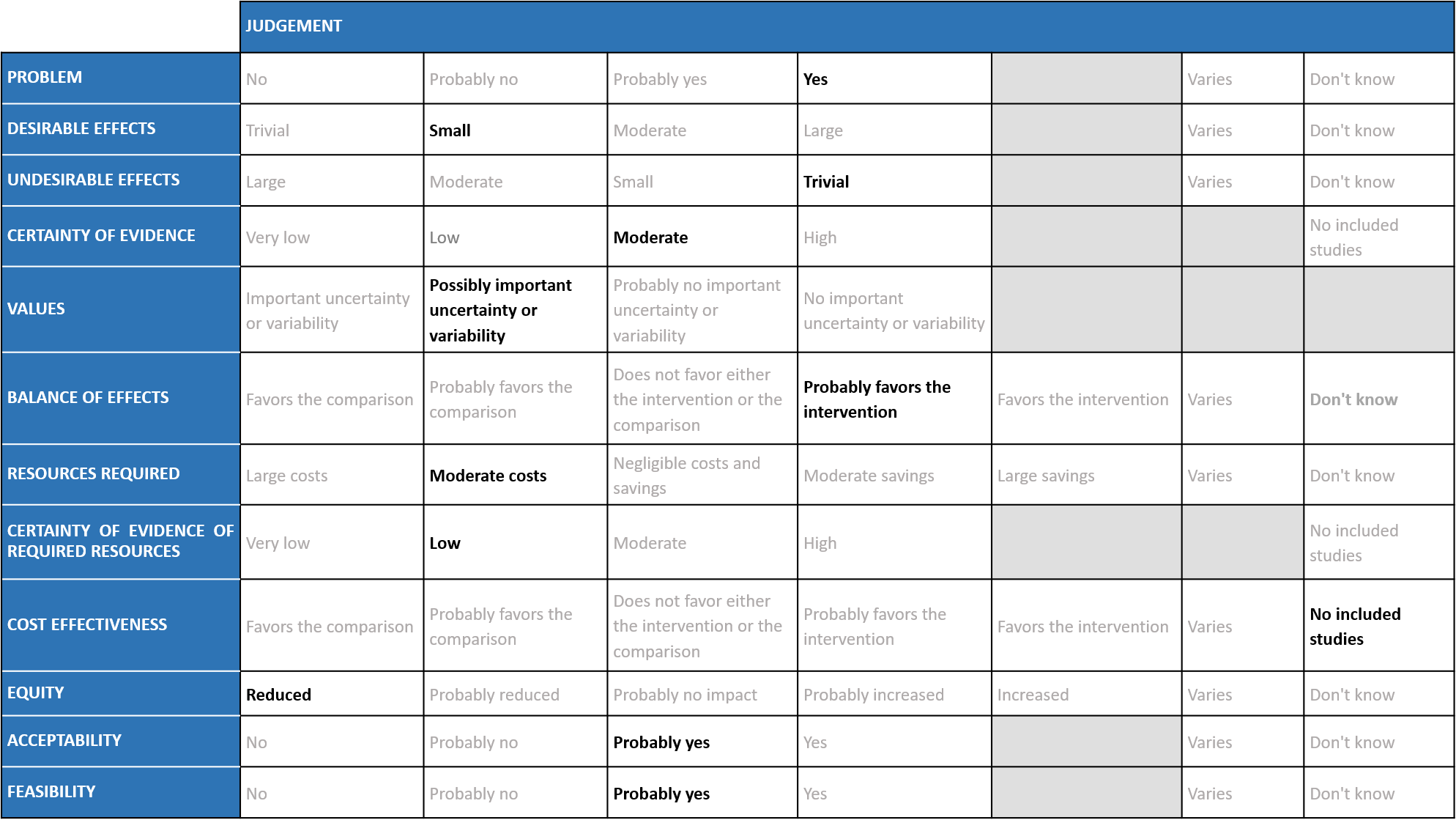
Problem
The COVID-19 pandemic in India with more than 43 million cases and over 0.52 million deaths has significantly impacted and stressed the health structure of the country. With a shortage of intensive care unit beds, oxygen and trained personnel the country is facing a major health crisis. This has prompted many irrational treatment interventions which are neither beneficial nor cost effective. Remdesivir, similar to other antivirals was believed to have a possible benefit in the early viral replication phase of the disease, and data now shows that there is a mortality reduction in mild to moderate COVID-19 disease in the out-patients. Most recently SOLIDARITY trial data has become available which provided useful information regarding its use in hospitalized patients with COVID-19 infection. However, it continues to be prescribed widely and the group judged the problem to be of the utmost priority.
Desirable effects
The group agreed that Remdesivir may have a beneficial effect on mortality and progression to oxygen therapy or non-invasive mechanical ventilation and they have noticed this personally even when treating patients. The group agreed there was a benefit in time to clinical improvement (2 point reduction in WHO ordinal scale) and clinical recovery (not requiring hospitalization for oxygen or medical care) when Remdesivir was compared with placebo/SOC, however the magnitude of this reduction in days was unclear.
The group also noted that the early trials did not use steroids as much as they are used now and the benefit of Remdesivir when steroids was on board was unclear in the past. In addition, the median duration of oxygen requirement was around 3 weeks in the ACTT-1 trial (8), which is quite prolonged compared to what we have seen more recently suggesting that treatment protocols have since evolved and we have data which now clearly suggests that there is a clearcut benefit across all clinical outcomes. This does have implications for a health system which is overwhelmed with a shortage of supplies and hospital beds. Taking these factors into consideration, overall the group members suggested a small to moderate benefit for Remdesivir.
Undesirable effects
Adverse events were not different between groups, nor were there adverse events leading to discontinuation and serious adverse events, suggesting this drug may be safe. The group noted evidence was lacking in pregnancy, renal and liver disease and overall judged the undesirable effects as trivial.
Certainty of evidence
The group felt that there was moderate to low certainty evidence for all outcomes, including those judged as critical.
Values
The group felt that there is possibly no important uncertainty or variability regarding values. In the initial stages, with the available data this result was unclear; however though the certainty of evidence was moderate regarding the effect estimate, the group agreed that stakeholders (for e.g., individual patient, health care providers, public or private health systems etc.) may value the beneficial effect of Remdesivir on these outcomes differently.
Balance of effects
The group discussed at length on many an occasion regarding the beneficial effects of Remdesivir. Data has evolved since the initial stages of the pandemic and now there is data to suggest that Remdesivir reduces mortality, time to clinical improvement/clinical recovery, progression to oxygen, non invasive or invasive mechanical ventilation. The group judged that at present with the evidence available the balance of effects seemed to favour the intervention, though the magnitude of benefit was moderate to low certainty of evidence. The group was clear they were not worried about adverse events. It is quite safe and has been extensively used in the country with no signal of harm. It could contribute to indirect benefits as it could free up the resources like hospital beds, oxygen supplies and impact patients' risk of nosocomial infections and other complications.
Resources required
The group felt that the costs were moderate. The group includes clinicians in different types of Indian hospitals who have a good idea of drug and hospitalization costs.
Certainty of evidence of required resources
No studies reporting this were reviewed by the group.
Cost effectiveness
The group noted the lack of data from available cost effectiveness studies relevant to the Indian context. There are only benefits in time to clinical improvement and progression to NIV/high flow oxygen which may result in early discharge and save hospital beds, supplies and costs; however this was low certainty evidence. This highlights the uncertainty regarding cost effectiveness that requires evidence from new clinical studies specific to the Indian context.
Equity
At this point in time this intervention would ‘reduce equity’ if found efficacious in Indian settings.
Acceptability
The group felt that this intervention is likely to have wide acceptance by all the relevant stakeholders (policy-makers, patients and clinicians), if further research proves its efficacy in the absence of increased adverse events.
Feasibility
This is a feasible intervention in the moderate to severe groups requiring oxygen as they are likely to need hospitalization by virtue of oxygen support.
Remdesivir should be considered in all hospitalized patients if cost and availability is not an issue. It should be given especially if in the early stages of the disease in all hospitalized patients. There is reduction in mortality, progression to oxygen therapy, non invasive and invasive ventilation as well as time to clinical improvement and recovery. Though this reduction in time to clinical improvement and recovery was seen, it was not quantifiable in number of days. However this does signal saving hospital resources for sicker patients. This effect seems to be over and above that seen with steroids.
Clinicians are familiar with this drug and wide use has revealed very low incidence of adverse effects. However, patients need to be given this drug by trained health care personnel either at a clinic or hospital or if at home by a skilled health care worker.
The group considered a potential subgroup effect across patients with early administration of Remdesivir.
(a) Oxygen requirement: For this analysis, the participants were stratified as: no supplemental oxygen requirement, low flow oxygen, high flow oxygen and non-invasive ventilation and invasive mechanical ventilation. In three RCTs (3, 8, 12) with 1332 participants, in the subgroup with low flow oxygen it did show a mortality benefit (RR 0.57; 95% CI 0.34 to 0.95).
(b)Early administration: Subgroup analysis to determine if Remdesivir benefits those patients who present early (<10 days) was considered, however only one study (9) addressed this and no mortality benefit was reported.
The committee had requested analyses of other important subgroups of patients including children, age > 50 years and those with co-morbidities, but unfortunately there was no data to address outcomes in these subgroups specifically.
New evidence suggests that Remdesivir is beneficial even in hospitalized patients with COVID-19 infection. Pharmacovigilance data over 2 years has suggested that this a safe drug with few adverse effects which should be easy to monitor even in an out-patient setting like bradycardia, hypotension and liver dysfunction. The certainty of evidence for any adverse events was low and for adverse events leading to drug discontinuation was very low, thus potential drug-related harm has still not been ruled out.
There is considerable uncertainty surrounding the benefits or harms of using Remdesivir in pre-specified subgroups such as immunocompromised, renal failure patients on dialysis, pregnant women and children. Hence there is a felt need to evaluate the efficacy of Remdesivir in the above sub groups.
- Eastman RT, Roth JS, Brimacombe KR, Simeonov A, Shen M, Patnaik S, et al. Remdesivir: A Review of Its Discovery and Development Leading to Emergency Use Authorization for Treatment of COVID-19. ACS Cent Sci. 2020 May 27;6(5):672–83.
- Frediansyah A, Nainu F, Dhama K, Mudatsir M, Harapan H. Remdesivir and its antiviral activity against COVID-19: A systematic review. Clin Epidemiol Glob Health. 2021;9:123–7.
- Wang M, Cao R, Zhang L, Yang X, Liu J, Xu M, et al. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020 Mar;30(3):269–71.
- Rochwerg B, Agarwal A, Zeng L, Leo YS, Appiah JA, Agoritsas T, et al. Remdesivir for severe covid-19: a clinical practice guideline. BMJ. 2020 Jul 30;m2924.
- Therapeutics and COVID-19: living guideline [Internet]. [cited 2022 May 11]. Available from: https://www.who.int/publications-detail-redirect/WHO-2019-nCoV-therapeutics-2022.3
- Remdesivir [Internet]. COVID-19 Treatment Guidelines. [cited 2022 May 11]. Available from: https://www.covid19treatmentguidelines.nih.gov/therapies/antiviral-therapy/remdesivir/
- Sharma DL. Clinical Guidance for Management of Adult COVID-19 Patients (Revised: 14/01/2022). :1.
- Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the Treatment of Covid-19 — Final Report. N Engl J Med. 2020 Oct 8;NEJMoa2007764.
- Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet Lond Engl. 2020;395(10236):1569–78.
- Spinner CD, Gottlieb RL, Criner GJ, Arribas López JR, Cattelan AM, Soriano Viladomiu A, et al. Effect of Remdesivir vs Standard Care on Clinical Status at 11 Days in Patients With Moderate COVID-19. JAMA. 2020 Sep 15;324(11):1–10.
- Mahajan L, Singh AP, Gifty. Clinical outcomes of using remdesivir in patients with moderate to severe COVID-19: A prospective randomised study. Indian J Anaesth. 2021 Mar;65(Suppl 1):S41–6.
- Ali K, Azher T, Baqi M, Binnie A, Borgia S, Carrier FM, et al. Remdesivir for the treatment of patients in hospital with COVID-19 in Canada: a randomized controlled trial. CMAJ. 2022 Feb 22;194(7):E242–51.
- Abd-Elsalam S, Salama M, Soliman S, Naguib AM, Ibrahim IS, Torky M, et al. Remdesivir Efficacy in COVID-19 Treatment: A Randomized Controlled Trial. Am J Trop Med Hyg. 2022 Mar 9;106(3):886–90.
- Ader F, Bouscambert-Duchamp M, Hites M, Peiffer-Smadja N, Poissy J, Belhadi D, et al. Remdesivir for the treatment of hospitalised patients with COVID-19: final results from the DisCoVeRy randomised, controlled, open-label trial [Internet]. medRxiv; 2022 [cited 2022 May 2]. p. 2022.03.30.22273206. Available from: https://www.medrxiv.org/content/10.1101/2022.03.30.22273206v2
- Remdesivir and three other drugs for hospitalised patients with COVID-19: final results of the WHO Solidarity randomised trial and updated meta-analyses. The Lancet. 2022 May;S0140673622005190.
- Repurposed Antiviral Drugs for Covid-19 — Interim WHO Solidarity Trial Results | NEJM [Internet].
Covid management Guidelines India Group- Anti-viral Working Group. Updated Remdesivir. Covid Guidelines India: Published online on June 16, 2022; URL: https://indiacovidguidelines.org/remdesivir-inpatient/
