Non-invasive ventilation (NIV) vs High flow nasal oxygen (HFNO) as the initial ventilatory strategy for COVID-19 Acute respiratory distress syndrome (ARDS)
This recommendation applies to acute COVID-19 in adults. Some of our recommendations vary according to the severity of COVID-19 illness. Definitions of the categories are based on the World Health Organization (WHO) criteria and can be viewed by clicking the plus (+) signs below.
RECOMMENDATION: We recommend using either Non-Invasive Ventilation (NIV) or high flow nasal oxygen (HFNO) as the initial ventilatory strategy in people with COVID-19 Acute Respiratory Distress Syndrome (ARDS) requiring ventilation.
DATE OF RECOMMENDATION: 27/7/2021
Definition of Severe illness
Pneumonia with ANY ONE of the following:
• severe respiratory distress or respiratory rate >30/min
• SpO2 <90% on room air
• NO invasive or non-invasive respiratory support needed
Definition of critical:
• Requirement for high-level respiratory support: noninvasive ventilation, high-flow oxygen (≥20 litres per minute) or invasive mechanical ventilation
• OR acute respiratory distress syndrome (PaO2/FiO2 ratio of <300)
• OR sepsis
• OR shock
Non-invasive ventilation (NIV) can reduce endotracheal intubation and mortality in patients with acute respiratory distress syndrome (ARDS)while preventing nosocomial complications in COVID-19 infection. NIV can be delivered via a face mask or helmet interface (a transparent hood that covers the entire head of the patient with a soft collar neck seal 1. High-flow oxygen therapy through a nasal cannula is a technique whereby heated and humidified oxygen is delivered to the nose at high flow rates. These high flow rates generate low levels of positive pressure in the upper airways, and the fraction of inspired oxygen (FIO2) can be adjusted by changing the fraction of oxygen in the driving gas.
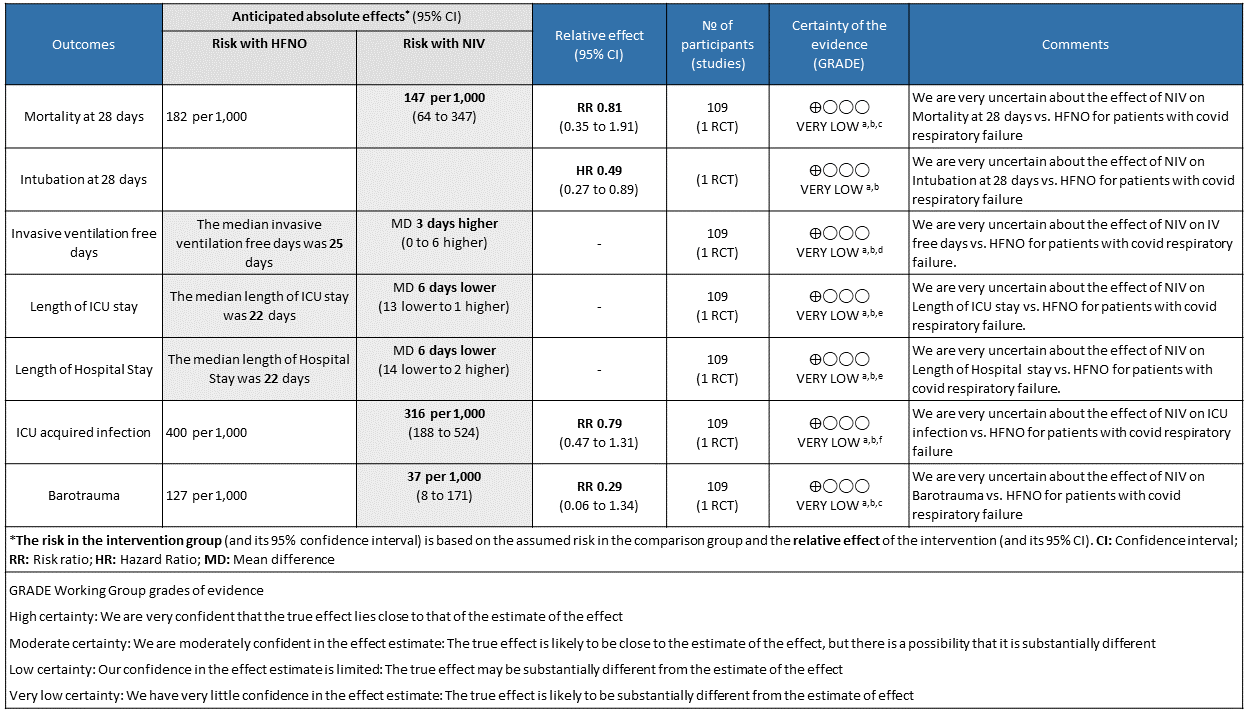
The evidence to answer the PICO question was very limited. There was only one randomized trial with 109 participants comparing NIV (delivered via helmet in the first 48 hours followed by HFNO), vs HFNO alone, as the initial ventilatory support for moderate to severe hypoxemic respiratory failure among COVID-19 patients. The study was small and the GRADING of the evidence very low, indicating that the research evidence is insufficient to recommend one over the other another as the initial ventilatory strategy.
The major issue with this trial was that patients in the NIV arm received NIV alone for only 48 hours; after which they also received HFNO in addition. However, the outcomes were recorded at 28 days. This makes it difficult to assess whether the outcomes reported in the NIV arm are related to NIV or the NIV followed by additional HFNO. The trial did not demonstrate a difference in the ‘number of days free of respiratory support’ within 28 days after enrolment. The number of ‘invasive ventilation free days’ was higher in the NIV group, but the overall rate of endotracheal intubation was lower in the NIV arm. No difference was demonstrated for mortality at 28 days, length of ICU stay, length of hospital stay, ICU acquired infection, and barotrauma in the two arms. Due to the methodological issue highlighted, and the small sample size, there is very uncertain evidence regarding the benefit of either modality over the other for these outcomes.
Date of latest search: 25th June 2021.
Date of completion & presentation to Expert Working Group: 25 June 2021.
Date of planned review: 25th December 2021.
Evidence synthesis team: Jefferson Daniel J, Avinash A Nair, Richard Kirubakaran, Priscilla Rupali and Bhagteshwar Singh.
Explanations
a. “Continuous infusion of sedative/analgesic drugs was administered to 20 patients (37%) in the helmet group and in 10 patients (18%) in the high-flow nasal oxygen group. Over the initial 48 hours of treatment, the mean (SD) Fio2 used in the helmet and high-flow nasal oxygen groups were 0.54 (0.12) and 0.58 (0.9), respectively. As per clinical decision, 32 patients (60%) in the high-flow nasal oxygen group vs 0 in the helmet group underwent prone position.” This would make the RoB2 high risk for domain 2, and therefore overall.
b. Downgraded by 2 levels for very serious indirectness; The data were from 1 trial in 1 high income country, Median severity of ARDS was higher than the usual severity at which NIV would be started, Helmets were used for NIV and the pressure settings were higher than usual maintenance settings used in most ICUs in India. Weaning off NIV in the first arm involved using HFNO, which is not followed in many ICUs in India.
c. Downgraded by 2 levels for imprecision due to few events and CI including important benefit and important harm.
d. Downgraded by 1 level for imprecision as CI crosses the clinical decision threshold between recommending and not recommending treatment.
e. Downgraded by 1 level for imprecision due to CI including no difference and important benefit.
f. Downgraded by 1 level for imprecision due to CI including important benefit and important harm with NIV.
The COVID-19 pandemic in India has caused considerable strain on the oxygen supply chain and ventilator manufacturers. Several treatment modalities have been tried for the management of respiratory failure and respiratory distress. Since the mortality rate was found to be higher in those who require invasive ventilation, appropriate management with non-invasive ventilation in the early phase of ARDS is of utmost importance (2). Continuous Positive Airway Pressure therapy (CPAP), Bilevel Positive Airway Pressure therapy (BPAP), Non-invasive ventilation mode on the ventilator and High Flow Nasal Cannula/Oxygen (HFNC/HFNO) are common non-invasive Respiratory Support (NRS), devices and are being used in various centres in India and abroad (3). Various interfaces for these ventilators have also been tried, such as helmet, oronasal mask and face mask(4).
The NIH guidelines (December 2020) and the Surviving Sepsis guidelines (March 2020) recommend the use of HFNO over NIV in cases of persistent respiratory failure despite conventional oxygen therapy (5,6). Their conclusion was based on the following two non-COVID data studies. In non-COVID acute hypoxemic respiratory failure, Jean-Pierre Frat JP et al. found that patients on HFNO had more ventilator-free days, lower 90-day mortality, and lower intubation rates than patients on NIV (7). A meta-analysis by Yue-Nan Ni et al. looked at the effectiveness of different oxygenation methods before intubation. They found that HFNO had a reduced rate of intubation and ICU mortality compared to NIV in patients with acute respiratory failure (8). Most other guidelines, including WHO, ERS, and the Australian National Guidelines, recommend either NIV or HFNO based on availability. Hence an equipoise exists as to whether NIV or HFNO would be the preferred initial ventilatory strategy in COVID19 ARDS. Each of these strategies have implications with regard to costs, specialised devices or equipment and optimum utilisation of oxygen as well as trained personnel to administer these modalities.
We searched the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Epistemonikos, and the COVID‐19‐specific resource www.covid‐nma.com for studies on this topic published up to 25 June 2021. Very few studies were found on this topic. Only 1 randomized control trial was available but we found many observational studies and a review article. Hence, we also included the latest Cochrane systematic review on Non-COVID acute hypoxic respiratory failure and the two studies (1 RCT and 1 Systematic review) based on which NIH guidelines were made.
We extracted data for the following outcomes, predefined by the Expert Working Group:
- Critical (primary for this review):
- All-cause mortality
- Need for intubation
- Important (secondary):
- Time to clinical improvement (WHO ordinal scale or other definitions)
- Length of stay in hospital
- Length of stay in critical care
- Duration of days free from invasive ventilation
- Adverse events:
- All
- Serious
- Nosocomial infections
- Pneumothorax/pneumomediastinum
- Discontinuation due to inability to tolerate therapy
Two reviewers independently assessed the eligibility of search results. One reviewer extracted data from the RCT study and assessed the risk of bias using the Cochrane Risk of bias (RoB) v2.0 tool. The two Non-COVID meta-analyses were evaluated with AMSTAR2. Later the whole team reviewed the ROB and AMSTAR and corrected discrepancies if any.
We used RevMan 5.4 to perform meta‐analysis using a random‐effects model for outcomes where pooling of effect estimates was appropriate. We used risk ratios (RR) for dichotomous outcomes and mean differences (MD) for continuous outcomes, with 95% confidence intervals (CIs). We used GRADE methodology to assess the certainty in the evidence and documented this in a 'Summary of findings table using GradeProGDT.
On advice from the methodology committee, we also summarised data from the non COVID-19 systematic reviews and non-randomized observational studies.
We looked at one RCT, 3 systematic reviews and 7 obeservational studies. (7–16) The RCT was a comparison between Helmet NIV vs HFNO. A conventional face mask NIV should be better than Helmet NIV due to less leak and better delivery of the pressure support. The RCT had a high risk of bias due to deviation from the intended interventions. More people in the HFNO group received proning and the need for IV sedation/analgesia was more in the NIV group. We reviwed information from three non-COVID ARDS related meta-analysis due to lack of data from COVID patients.
Out expert working group classified all cause mortality and need for intubation as critical outcomes. Time to clinical improvement, length of stay in hospital and ICU, duration of days free from invasive ventilation, nosocomial infections and barotrauma were other important outcomes considered. However we had only one RCT addressing most of the above outcomes.
The HENIVOT Trial
The HENIVOT Trial was the only RCT on this topic published so far. (8) It was a multicentre RCT done in 4 ICUs in Italy. The study compared Helmet NIV with high pressures of PS 10-12 cm of H20 and PEEP of 10-12 cm of H2O against HFNO 60L/min for at least two days. The patients in both arms were comparable and had no risk of bias at randomization. The NIV arm patients received HFNO after two days once they met predefined criteria for improvement. There was deviation of protocol on account of sedation/analgesia infusion (more in NIV) and proning (more in HFNO) compared to the other arm. Proning is an independent confounding factor for improvement and may tilt the evidence in favour of HFNO.
Critical outcomes
a. All-cause mortality at 28 days
Very low certainty of evidence revealed that there was an uncertain effect of NIV over HFNO for the outcome of all-cause mortality at day 28; RR=0.81 (95% CI 0.35-1.81). The in-hospital mortality was 13 (24%) in the NIV arm compared to 14 (25%) in the HFNO arm, with an absolute difference of 1 death. This was not considered statistically or clinically different.
b. Need for intubation.
Very low certainty evidence revealed that intubations were lower (intubation at 28 days) in the Helmet NIV group (28%) compared to HFNO (51%), HR = 0.49 (95% CI, 0.27-0.89 and this was statistically significant with a p value = 0.02).
Important Outcomes
a. Length of stay in hospital
Very low certainty of evidence revealed that the length of stay in hospital was lower in the NIV group with a mean difference of 6 days (14 lower to 2 higher). In addition, median (IQR) was 21 (14 to 30) in the NIV group as compared to the HFNO group 22 (13 to 44) and seemed that the median was not statistically or clinically significant.
b. Length of stay in critical care
Very low certainty of evidence revealed a very uncertain effect of NIV as compared to HFNO with regard to ICU stay but the mean difference was 6 days (1-13 days). When expressed as median (IQR), the length of stay in ICU was lower in the NIV group 9 (4 to 17) [compared to the HFNO group 10 (5 to 23), but was not statistically or clinically significant.
c. Duration of days free from invasive ventilation
Very low certainty evidence revealed an uncertain effect of NIV vs HFNO with regard to invasive ventilation free days mean difference = 3 days (0-6 days). Invasive ventilation free days at 28 days was higher in the NIV group 28 days (13 to 28) [median (IQR), d] compared to the HFNO group 25 days (4 to 28). This mean difference of 3 days was both clinically and statistically significant (p = 0.04)
d. Nosocomial infections
Very low certainty of evidence revealed an uncertain effect of NIV vs HFNO in patients with COVID-19 ARDS; RR = 0.79 (0.47-1.31). Numerically 22 (40%) patients in the HFNO group acquired nosocomial infections compared to the NIV group 17 (31%), but this was not statistically significant.
e. Pneumothorax/pneumomediastinum
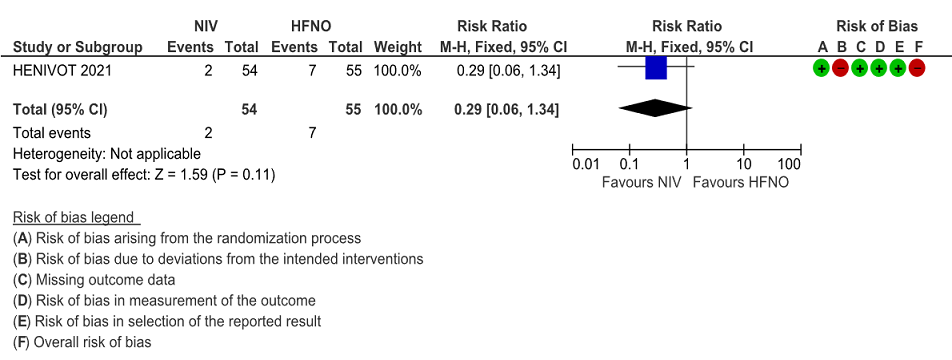
Very low certainty of evidence revealed that barotrauma was not significantly different between NIV (4%) and HFNO (13%) with RR=0.29(95% CI 0.06-1.34). When subcutaneous emphysema was evaluated, it was slightly more in the HFNO group [0% NIV vs 9% HFNO, p= 0.06].
This suggested that NIV via helmet with high pressure (PS) or Positive end expiratory pressure (PEEP) was better than HFNO at 60L/min in reducing intubation rate and ventilator-free days in moderate to severe ARDS.
In conclusion, NIV was found comparable to HFNO in terms of mortality, hospital stay and adverse events. Though intubations and invasive ventilation free days were secondary outcomes in the HENIVOT trial, they were found to be adequately powered on retrospective calculations. This suggested that NIV via helmet with high pressure (PS) or Positive end expiratory pressure (PEEP) was superior to HFNO at 60L/min in reducing intubation rate and ventilator-free days in moderate to severe ARDS. However, the major limitation with this trial was that patients in the NIV arm received NIV alone for only 48 hours; after which they also received HFNO in addition with the outcomes being recorded at 28 days. Hence it was difficult to assess whether the outcomes in terms of benefits/harm reported in the NIV arm were related to NIV or the NIV followed by additional HFNO.
1. Non-COVID Systematic reviews
Two Non-COVID-19 systematic reviews were considered due to the shortage of literature comparing these two modalities in the COVID-19 era(2,10). Lewis SR et al. included RCTs up to April 2020 that looked at NIV vs HFNO in non-COVID-19 ARDS (10). This Cochrane review also included patients who were initiated on NIV/HFNO after being extubated from invasive mechanical ventilation. They found no significant difference between NIV and HFNO in terms of escalation of therapy, intubation and in-hospital mortality. Respiratory infection was also no different in each of type of respiratory support group or HFNO groups RR 0.51 (0.17 to 1.52). There was a subgroup analysis for treatment failure in patients who had no prior intubations vs those previously intubated, which also showed no difference between high flow nasal oxygen vs non-invasive respiratory support which included non-invasive positive pressure ventilation (NIPPV), continuous positive airway pressure (CPAP) and bilevel positive airway pressure (biPAP) in both the groups. Ferreyro BL et al. performed a network meta-analysis, that compared conventional oxygen therapy, facemask NIV, helmet NIV and HFNO(11). This non-COVID-19 ARDS study showed no difference between Face mask NIV vs HFNO in terms of all-cause mortality and intubation.
2. Non-randomized observational studies.
Crimi C et al. analyzed observational studies on COVID and Non-invasive respiratory support published till April 2020. The pooled statistics showed that the overall failure rate of NRS varied between 52% to 92%. The mean utilization rate was 31% for HFNO and 30% for NIV across the studies. There were 39 observational studies identified in our literature search, of which 14 had data for our predefined outcomes as per our PICO. Six of these studies compared HFNO and NIV to some extent in certain aspects, but they were non-randomized trials. The outcomes of interest from these studies are tabulated as follows. A direct comparison is not possible as the NIV/HFNO protocols used in these centres may vary widely. We were also unable to determine which interface was used to deliver cPAP or NIV in most studies. Some centres which lack experienced manpower in running NIVs preferred using HFNO. Timing and criteria of intubation also varied between various centres. On closer observation of the table, despite all the above mentioned drawbacks, NIV and CPAP are very much comparable to each other in terms of intubation and mortality.
Table 1 Non-randomized observational Studies
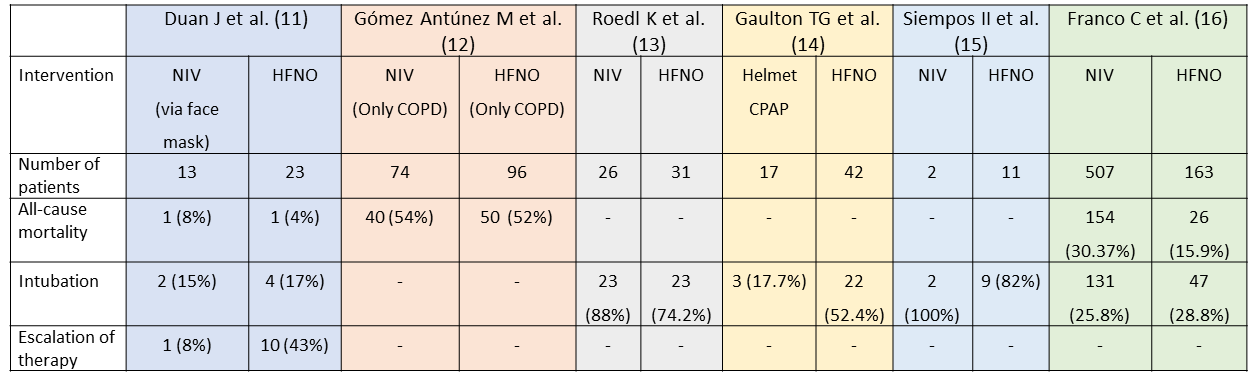
Figure 1: Mortality at 28 days
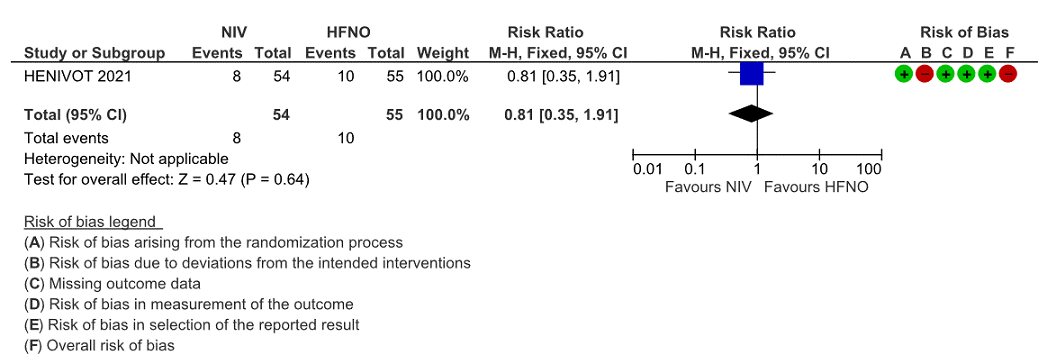
Figure 2: Intubation at 28 days
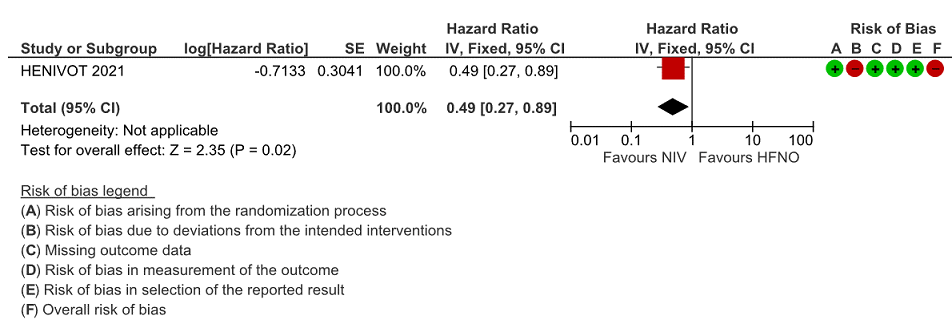
Figure 3: Intubation at 4, 8 and 28 days
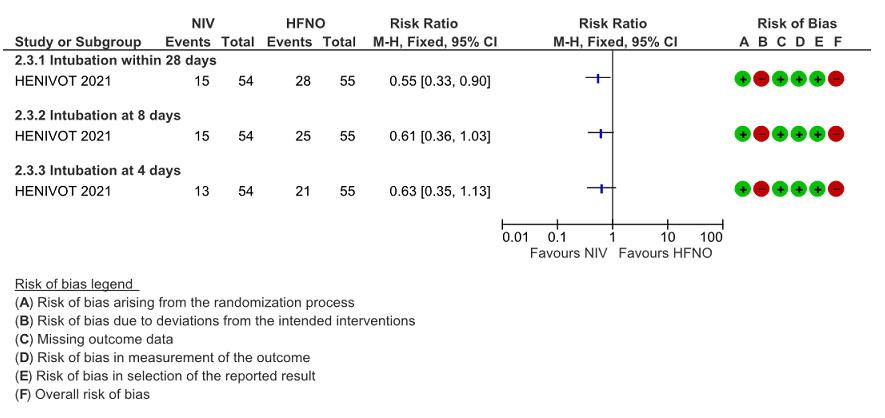
Figure 4: Invasive ventilation free days
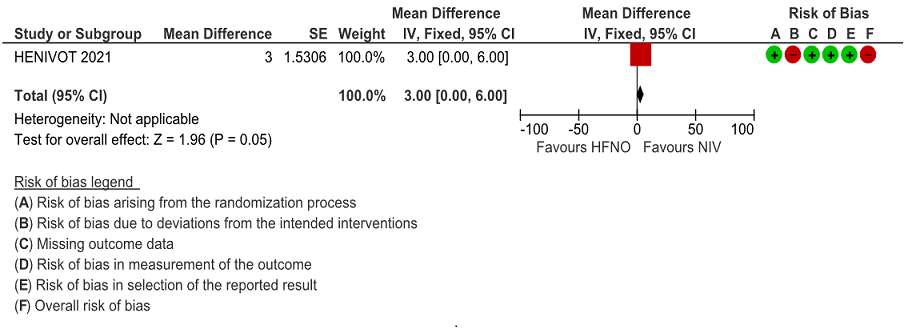
Figure 5: Length of ICU stay
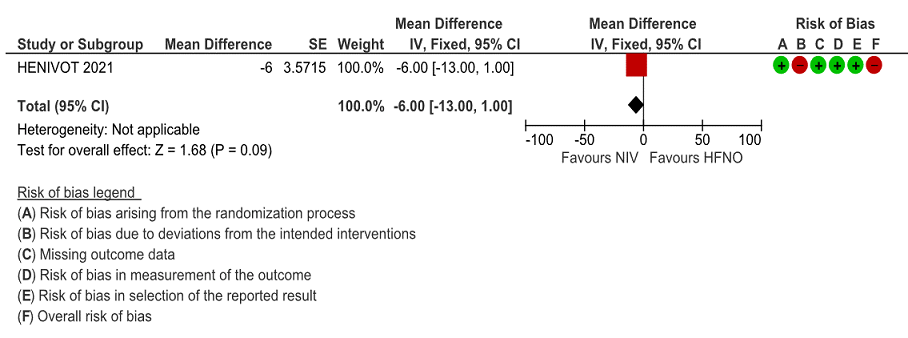
Figure 6: Length of Hospital Stay
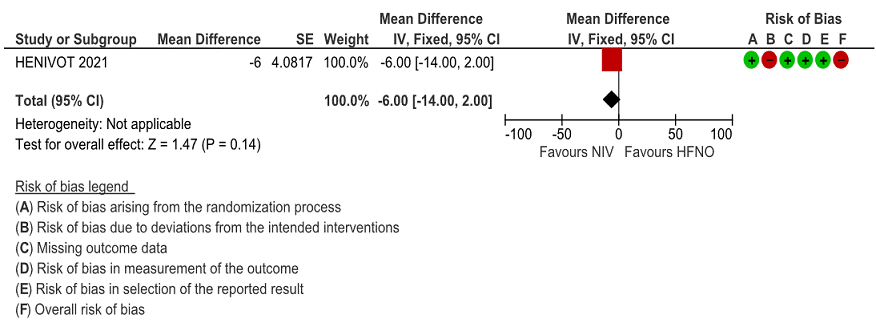
Figure 7: ICU Acquired infection
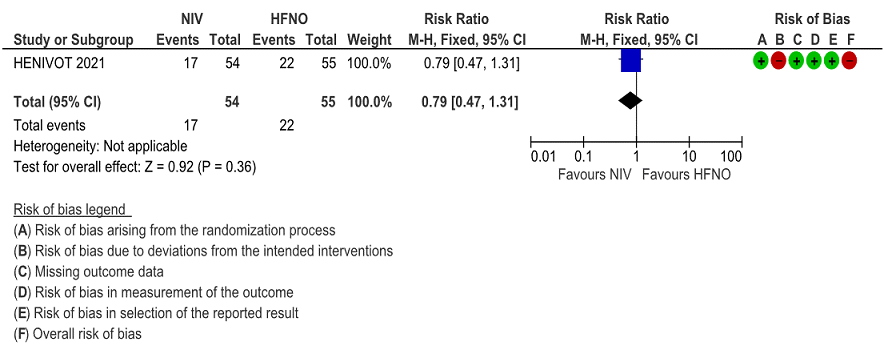
Figure 8: Barotrauma
Problem
The key healthcare challenge of the COVID-19 pandemic has been the safe delivery of respiratory support on a large scale for management of the resultant acute respiratory distress syndrome (ARDS). Need for low technology and easily implementable oxygen delivery or respiratory support interventions to prevent further progression of ARDS has become the need of the hour. However, HFNO though used widely during this pandemic its efficacy vs NIV has been an area of equipoise and has led to much debate, with regard to the benefits with prevention of progression to invasive mechanical ventilation and harms to healthcare workers due to aerosol generation. NIV is resource intensive and needs a specific skill set which is not easy to come by, while HFNO is likely to be less resource and personnel intensive. Another important point to consider has been which intervention is likely to result in lower consumption of oxygen considering the acute urgency of the situation.
Desirable effects
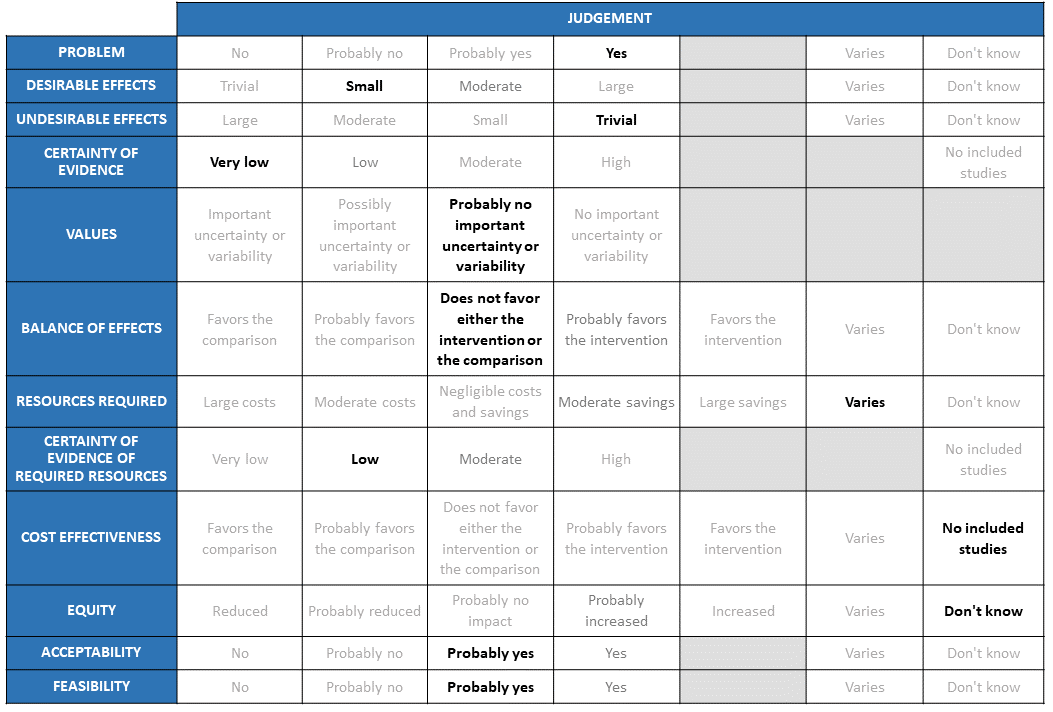
The group considered the evidence presented. The previous meeting had considered evidence in the pre-covid era and with the results available from the HENIVOT trial the group decided to evaluate, evidence in the COVID era only. The evidence seemed to favour NIV in the outcomes of intubation at 28 days and various time to event outcomes like ventilation free days (invasive ventilation), length of ICU and hospital stay (by 1-14 days). It was not significant for outcomes like mortality, intubation at 4 and 8 days and adverse outcomes like ICU related nosocomial infections and barotrauma. The group also discussed at length the indirectness of this low certainty evidence there were many deviations from normal clinical practice (1) the various problems within the HENIVOT trial of NIV arm being given NIV only for 48 hours prior to switching to HFNO which may not again give a clear picture with regard to benefit of NIV over HFNO (2) Switching from HFNO directly to invasive mechanical ventilation without a trial of NIV (3) NIV was delivered by helmets which is not a common interface used in India. Hence the consensus in the group was that the evidence for desirable effects of NIV over HFNO was small.
Undesirable effects
The undesirable effects of NIV were thought to be trivial. NIV when delivered through a helmet did not seem to offer an advantage or cause a disadvantage when compared with HFNO for the outcomes of ICU infections or barotrauma. There was a concern regarding the possibility of nosocomial transmission of COVID-19 if given HFNO, though this risk is still not negligible if NIV delivered via mask rather than through a helmet.
Certainty of evidence
This was agreed upon as “very low” based on the evidence presented.
Values
The group agreed that there was no important uncertainty or variability with regard to the outcomes studied. These were valid outcomes which were clinically relevant.
Balance of effects
The magnitude of desirable effects being small and undesirable effects being very low for the intervention of NIV and comparison of HFNO with very low certainty of evidence the group voted that the balance of effects did not favour either the intervention or the comparison.
Resources required
The group discussed that the costs varied depending on the interface, skill set of personnel involved, amount of oxygen utilised and the setting that it was used in. Helmets use far more oxygen as compared to the masks used for NIV, however the group agreed that some of that could be balanced by using air rather than oxygen. Masks are also a less expensive alternative as compared to helmets. There may be small savings if we use helmet with air rather than oxygen.
Certainty of evidence of required resources
The group considered this as low but cautioned that this was based on clinical experience rather than studies.
Cost effectiveness
Cost effectiveness would depend on resources like the interfaces, oxygen flow rate, devices and capital infrastructure and in addition will differ based on provider and health care setting. In lower resource settings, the costs are significantly in favour of NIV use. However, equipment is different and the flow rates can vary from 30L to 70L of oxygen/minute. Masks can also vary in cost. They felt that though HFNO was probably technically easier to administer through specialised devices, NIV may require ventilators suited to delivering this modality of oxygen along with the required setting and skilled personnel to deliver this. However, it would indeed be ideal to have studies which answer this question of cost effectiveness. Hence the group voted for no included studies for this section.
Equity
The group felt unable to comment on the impact for equity. The panel discussed that HFNO requires less expertise, but requires a specific device to implement. Capital costs differ if NIV used via ventilator/dedicated NIV machine. Hence the group voted “don’t know” for the impact on equity.
Acceptability
The group agreed that NIV would indeed acceptable. However, there are likely to be some differences with regard to implementation, experience and comfort level at each of the centres. NIV via any interface would be acceptable to clinicians, if resources and expertise were available. However, for patients or hospital administrators this is less clear, but some pre-covid studies suggest acceptability to patients.
Feasibility
The panel felt that in health facilities with a physician or anaesthetist, and the requisite devices and medical oxygen/air at required flow rates, this is feasible. This may vary between settings, but patients being considered for this intervention are likely to be in an appropriate setting. Members of the panel have used NIV in lower resource/remote settings.
Adequate training and devices are needed to implement either of these interventions. HFNO often considered as first step up from low-flow O2 with then NIV considered as the next step, but group acknowledged that O2 availability can be an issue. The group concluded that the use of NIV and HFNO are quite dependent on the end user as well as resources available. Majority of clinicians are familiar with NIV, and its use may be preferred when oxygen availability is a concern. HFNO is a newer respiratory support therapy which may not be available at most centres. In addition, it utilizes more oxygen especially at higher flow rates and is easier to use (needs less training than NIV). Often these therapies can be combined for a patient, with HFNO as a step up from low flow O2 and step down from NIV. Overall, NIV seemed the option that is familiar and beneficial. However, if equipment and trained personnel are not in place, High Flow Nasal Oxygen may be used.
Mild (PF ratio 200-300) vs moderate ARDS (PF 100- 200): No subgroup analysis was done in the only RCT evaluated. Given that the use of NIV or HFNO did not result in statistically different outcomes, either can be used for moderate ARDS. In mild COVID-19 ARDS, no specific recommendation can be made based on current evidence. Perhaps decisions need to be taken based on individual patient’s condition and response to therapy, resources and experience of clinicians attending to the patient.
No specific recommendations can be made with regard to the following disease states based on available evidence-
- Pre-existing COPD
- Obesity
- Co-existing cardiac failure (acute/chronic)
- Age > 65 years
Although currently there is no evidence to support NIV over HFNO or vice versa as the initial mode of respiratory support in mild or moderate ARDS, this will be revisited as new evidence emerges. Large randomised studies where patients received exclusively one of these interventions against the other will be monitored periodically. In addition, studies which use oronasal masks as the interface in the non-invasive ventilation group will be more practically applicable in India, due to lower cost, lower oxygen use and ease of administration. Evidence of side effects of both these modalities needs monitoring in studies with larger number of patients, to see if one is safer than the other.
The group felt that we needed randomized controlled trials with regard to the following
• RCT comparing NIVwith HFNO with clearly defined outcomes like mortality and requirement of IMV in early ARDS.
• RCT comparing different interfaces face mask, helmets are also research priorities as there are no head to head comparisons available.
- Patel BK, Wolfe KS, Pohlman AS, Hall JB, Kress JP. Effect of Noninvasive Ventilation Delivered by Helmet vs Face Mask on the Rate of Endotracheal Intubation in Patients With Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA. 2016;315(22):2435-2441. doi:10.1001/jama.2016.6338.
- Lim ZJ, Subramaniam A, Ponnapa Reddy M, Blecher G, Kadam U, Afroz A, et al. Case Fatality Rates for Patients with COVID-19 Requiring Invasive Mechanical Ventilation. A Meta-analysis. Am J Respir Crit Care Med. 2021 Jan 1;203(1):54–66.
- Crimi C, Noto A, Cortegiani A, Impellizzeri P, Elliott M, Ambrosino N, et al. Noninvasive respiratory support in acute hypoxemic respiratory failure associated with COVID-19 and other viral infections. Minerva Anestesiol. 2020 Nov;86(11):1190–204.
- Patel BK, Kress JP, Hall JB. Alternatives to Invasive Ventilation in the COVID-19 Pandemic. JAMA. 2020 Jul 7;324(1):43–4.
- COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. [Internet]. [cited 2021 Jun 3]. Available from: https://www.covid19treatmentguidelines.nih.gov/critical-care/oxygenation-and-ventilation/.
- Alhazzani W, Evans L, Alshamsi F, Møller MH, Ostermann M, Prescott HC, et al. Surviving Sepsis Campaign Guidelines on the Management of Adults With Coronavirus Disease 2019 (COVID-19) in the ICU: First Update. Crit Care Med. 2021 Mar;49(3):e219.
- Frat JP, AW Thille, Mercat A etal. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med 2015; 372:2185-2196
DOI: 10.1056/NEJMoa1503326. - Ni YN, Luo J, Yu H, Liu D, Ni Z, Cheng J, Liang BM, Liang ZA. Can High-flow Nasal Cannula Reduce the Rate of Endotracheal Intubation in Adult Patients With Acute Respiratory Failure Compared With Conventional Oxygen Therapy and Noninvasive Positive Pressure Ventilation?: A Systematic Review and Meta-analysis. Chest. 2017 Apr;151(4):764-775. doi: 10.1016/j.chest.2017.01.004. Epub 2017 Jan 13. PMID: 28089816.
- Grieco DL, Menga LS, Cesarano M, Rosà T, Spadaro S, Bitondo MM, et al. Effect of Helmet Noninvasive Ventilation vs High-Flow Nasal Oxygen on Days Free of Respiratory Support in Patients With COVID-19 and Moderate to Severe Hypoxemic Respiratory Failure: The HENIVOT Randomized Clinical Trial. JAMA. 2021 May 4;325(17):1731–43.
- Lewis SR, Baker PE, Parker R, Smith AF. High-flow nasal cannulae for respiratory support in adult intensive care patients. Cochrane Database Syst Rev. 2021 Mar 4;3:CD010172.
- Ferreyro BL, Angriman F, Munshi L, Del Sorbo L, Ferguson ND, Rochwerg B, et al. Association of Noninvasive Oxygenation Strategies With All-Cause Mortality in Adults With Acute Hypoxemic Respiratory Failure: A Systematic Review and Meta-analysis. JAMA. 2020 Jul 7;324(1):57–67.
- Duan J, Chen B, Liu X, et al. Use of high-flow nasal cannula and noninvasive ventilation in patients with COVID-19: A multicenter observational study [published online ahead of print, 2020 Jul 29]. Am J Emerg Med. 2020;S0735-6757(20)30666-5. doi:10.1016/j.ajem.2020.07.071.
- Gómez Antúnez M, Muiño Míguez A, Bendala Estrada AD et al. , SEMI-COVID-19 Network. Clinical Characteristics and Prognosis of COPD Patients Hospitalized with SARS-CoV-2. Int J Chron Obstruct Pulmon Dis. 2021 Jan 5;15:3433-3445. doi: 10.2147/COPD.S276692. PMID: 33447021; PMCID: PMC7801905.
- Roedl K, Jarczak D, Thasler L, Bachmann M, Schulte F, Bein B, et al. Mechanical ventilation and mortality among 223 critically ill patients with coronavirus disease 2019: A multicentric study in Germany. Aust Crit Care Off J Confed Aust Crit Care Nurses. 2021 Mar;34(2):167–75.
- Gaulton TG, Bellani G, Foti G, Frazer M, Fuchs BD, Cereda M. Early Clinical Experience in Using Helmet Continuous Positive Airway Pressure and High-Flow Nasal Cannula in Overweight and Obese Patients With Acute Hypoxemic Respiratory Failure From Coronavirus Disease 2019. Crit Care Explor. 2020 Sep;2(9):p e0216.
- Siempos II, Xourgia E, Ntaidou TK, et al. Effect of Early vs. Delayed or No Intubation on Clinical Outcomes of Patients With COVID-19: An Observational Study. Med., 23 December 2020. https://doi.org/10.3389/fmed.2020.614152.
- Franco C, Facciolongo N, Tonelli R, et al. Feasibility and clinical impact of out-of-ICU noninvasive respiratory support in patients with COVID-19-related pneumonia. Eur Respir J. 2020;56(5):2002130. Published 2020 Nov 5. doi:10.1183/13993003.02130-2020.
Covid Management Guidelines India Group - Respiratory Therapy Working Group. Non-Invasive Ventilation (NIV) Vs High Flow Nasal Oxygen (HFNO). Covid Guidelines India; Published online on July 27, 2021; URL :- https://indiacovidguidelines.org/niv-vs-hfno/ (accessed
